Parents prepare for COVID-19 vaccine for teens
FOX 2 - On Monday, the U.S. Food and Drug Administration Monday expanded emergency use authorization of the Pfizer COVID-19 vaccine in the United States for older children ages 12 to 15. But what does that mean for parents of teenagers?

Vaccine rollout prep is underway for teens
On Monday, the U.S. Food and Drug Administration on Monday expanded emergency use authorization of the Pfizer COVID-19 vaccine in the United States for older children ages 12 to 15.
Katrina Ishioka is 14 and has been waiting for the vaccine to be available for the same reason as everyone else - she wants her life back.
"I really want things to get back to as normal as they can be," Katrina said.
She's been patiently waiting and could get it as soon as this Friday. Her mom, Naomi, oversees the COVID-19 vaccine clinic at Troy Beaumont.
"All but Katrina here were vaccinated already we have been waiting for this moment to get her vaccinated," Naomi said.
Now that the FDA has authorized it, an advisory committee at the Center for Disease Control will meet on Wednesday to decide whether to approve the authorization.
The next step, Naomi says, is like it was back in December when it was first approved: logistics. Including keeping track and document vaccinations for kids.
"For people our age the best thing we can do is prevent things from happening and it's going to make everything better in the end," Katrina said.
Naomi said she made her daughter part of the vaccine conversation and says part of the problems she's seen is that kids in this age group are getting a lot of misinformation about vaccines.
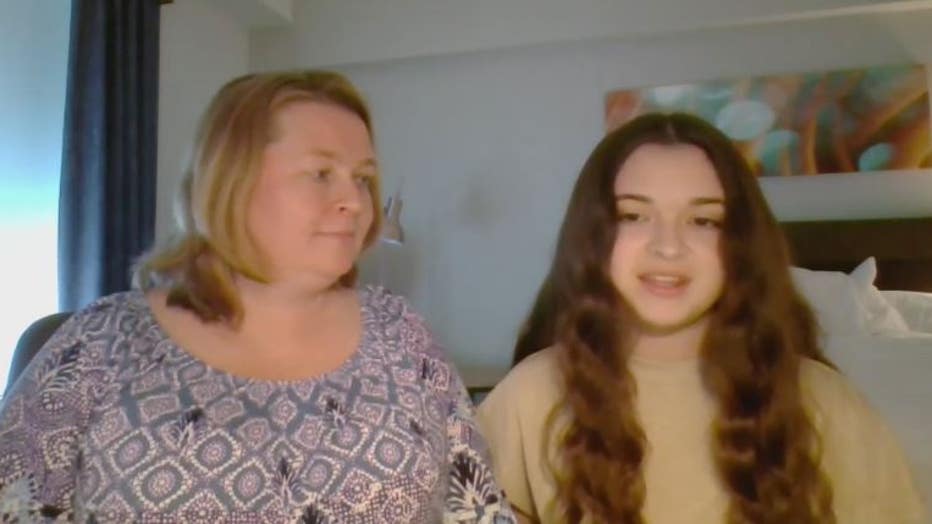
"I'm really proud of her for understanding that the science is good and the research shows it's safe and very effective," Naomi said. "If you just see it on social media and you don't see it in mainstream media, there is a very low likelihood that it's true. If you're only hearing about it from some influencer on TikTok it might not be true."
Pfizer reports that its COVID-19 vaccine was 100% effective at preventing the virus in children in this age group and has already started testing in children between 2 and 11.
Experts say children must get the shots if the country is to vaccinate the 70% to 85% of the population necessary to reach what's called herd immunity.

